 
Since sulfur has six valence electrons, we conclude that two electrons are not involved in the bonding, i.e., that there is a lone pair. In the sulfite ion, SO 3 2– for example, the oxidation number of sulfur is +4, suggesting that only four sulfur electrons are involved in the bonding. This changes the formula to 3- (0+4), yielding a result of -1. Boron (B) possesses three valence electrons, zero non-bonded electrons, and four bonds around it. Oxidation numbers can sometimes also be useful in writing Lewis structures, particularly for oxyanions. Formal Charge (number of valence electrons in neutral atom)- (non-bonded electrons + number of bonds) Example 1: Take the compound BH4 or tetrahydrdoborate. The formula for calculating formal charge is shown below: Formal Charge valence e- ( non-bonding e- + ½ bonding e- ) Note: the formal charges must add up to the net charge of the molecule also, like formal charges cannot appear next to each other within the molecule. This type of reaction can be recognized because it involves a change in oxidation number of at least one element. The first part is the number of valence electrons the atom donates to the Lewis dot Structure. Oxidation numbers are mainly used by chemists to identify and handle a type of chemical reaction called a redox reaction, or an oxidation-reduction reaction. Calculating Formal Charge: The following equation determines the formal charge for each atom in a molecule or polyatomic ion. please use regional emissions factors available in AVERT or eGRID.\,\] Note that the calculator uses national average emissions factors for electricity, which may not be accurate for your region. For electricity consumption, the calculator uses an average emissions factor that includes both baseload and non-baseload generation. Studied the topic name and want to practice Here are some exercises on Lewis Theory of Chemical Bonding practice questions for you to maximize your. * The Equivalencies Calculator uses different emissions factors for electricity depending on whether it is avoided or consumed at typical scales, energy efficiency and renewable energy programs and projects do not affect baseload power generation, so the calculator uses a non-baseload emissions factor. For more accurate estimates, please use regional emissions factors available in AVERT or eGRID. 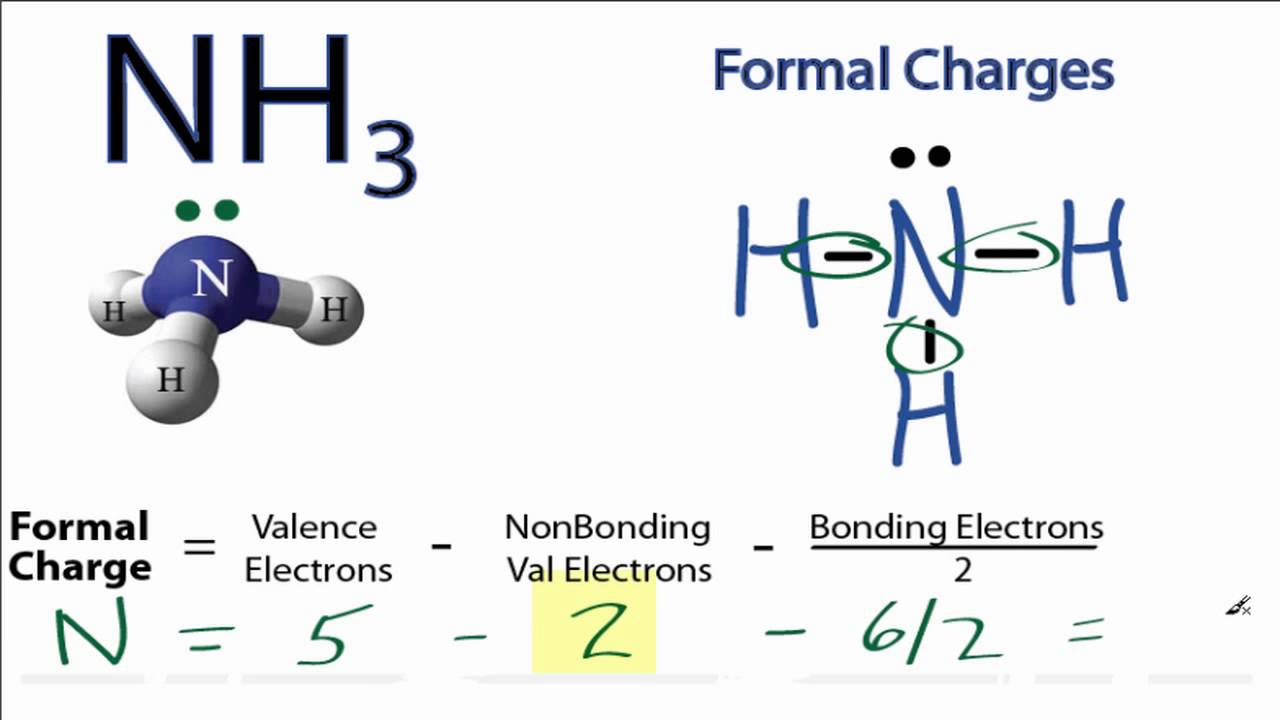
Another way of saying this is that formal charge results when we take the number of valence electrons of a neutral atom, subtract the nonbonding. Kilowatt-hours used Choose kilowatt-hours used when entering data on electricity use, such as your household’s or company’s annual electricity consumption. The formal charge of an atom in a molecule is the hypothetical charge the atom would have if we could redistribute the electrons in the bonds evenly between the atoms. Mathematical Method Formal charge equation is based on the comparing the number of electrons in the individual atom with that in the structure. The Formal Charge is defined by the relationship: Formal Charge number of valence electrons in an isolated atom - (number of lone pair electrons) + ½ (number of bonding electrons) With the definitions above, we can calculate the Formal Charge on the thiocyanate Ion, SCN -: Table 7.13. 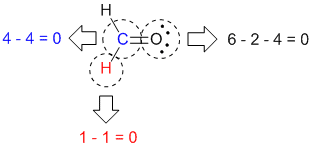
Holidays and weekends are included in the calculation. 
For more accurate estimates, please use regional emissions factors available in AVERT or eGRID. Other forums for resolution may be pursued at the same time as the processing of the EEOC charge. Note that the calculator uses national average emissions factors for electricity, which may not be accurate for your region. The formal charge of an atom in a molecule is the hypothetical charge the atom would have if we could redistribute the electrons in the bonds evenly between the atoms. Kilowatt-hours avoided Choose kilowatt-hours avoided when entering data on electricity use avoided through energy efficiency or fossil fuel electricity generation avoided through renewable energy. To see the methodology used to determine annual greenhouse gas emissions per passenger vehicle, visit the Calculations & References page for equations and sources used. A mathematical formula, a diagram, and the instinctive method are three different ways to calculate the formal charge. For the calculator’s purposes, passenger vehicles are defined as 2-axle 4-tire vehicles, including passenger cars, vans, pickup trucks, and sport/utility vehicles. The oxidation number of each atom can be calculated by subtracting the sum of lone pairs and electrons it gains from bonds from the number of valence electrons. Gasoline-powered passenger vehicles While passenger vehicles are not a unit of energy consumption, they do consume energy. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
